Transient absorption of DCM in ethanol
12/12/2018Transient absorption of DCM dye (4-
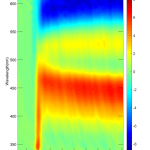
Results show positive signal centered around 450nm corresponding to overlap of higher state absorption and a ground state bleach with higher state absorption being of greater value. Negative band centered around 600nm corresponds to stimulated emission. During the first few hundreds of femtoseconds positive signal at lower wavelengths is present due to transient absorption artefacts (2-photon absorption, cross-phase modulation, etc.).
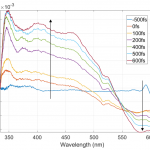
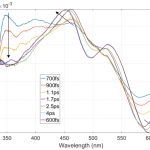
The figures of associated spectra show time-shift (black arrows) of positive and negative bands with positive band shifting towards blue wavelengths and negative band towards red wavelengths. Time-shift is present because of the solvation (solvent relaxation). With ethanol being polar solvent and DCM molecule having increased dipole moment value in its 1st excited state solvent molecules are reorienting to lower the energy of the 1st photoexcited state, thus increasing the energy required for higher state absorption and lowering the energy of stimulated emission.
 Group for Applied Ultrafast Spectroscopy
Group for Applied Ultrafast Spectroscopy